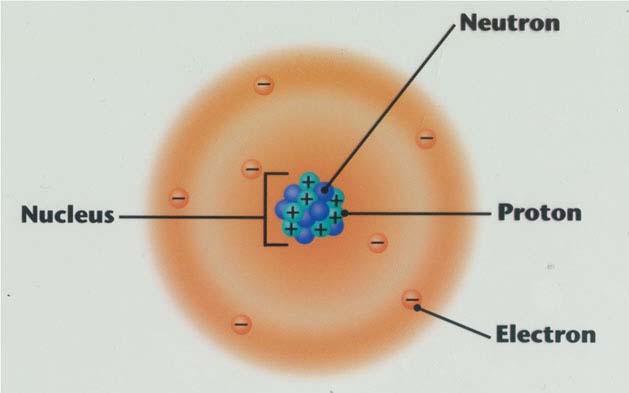
Much of an atom's positive charge is concentrated in a relatively tiny volume at the center of the atom, known today as the nucleus.The atom's electron cloud does not influence alpha particle scattering.Thus, Rutherford did not formally suggest the two numbers (periodic table place, 79, and nuclear charge, 98 or 100) might be exactly the same.Ī month after Rutherford's paper appeared, the proposal regarding the exact identity of atomic number and nuclear charge was made by Antonius van den Broek, and later confirmed experimentally within two years, by Henry Moseley. However, Rutherford did not attempt to make the direct connection of central charge to atomic number, since gold's "atomic number" (at that time merely its place number in the periodic table) was 79, and Rutherford had modelled the charge to be about +100 units (he had actually suggested 98 units of positive charge, to make half of 196). For gold, this mass number is 197 (not then known to great accuracy) and was therefore modelled by Rutherford to be possibly 196 u. The Rutherford paper suggested that the central charge of an atom might be "proportional" to its atomic mass in hydrogen mass units u (roughly 1/2 of it, in Rutherford's model). But in actuality the Northern Irish physicist, Joseph Larmor, created the first solar system model of the atom in 1897. Jean Baptiste Perrin claimed in his Nobel lecture that he was the first one to suggest the model in his paper dated 1901. Thomson also had rings of orbiting electrons. It did mention the atomic model of Hantaro Nagaoka, in which the electrons are arranged in one or more rings, with the specific metaphorical structure of the stable rings of Saturn. The Rutherford model served to concentrate a great deal of the atom's charge and mass to a very small core, but did not attribute any structure to the remaining electrons and remaining atomic mass. This was in a gold atom known to be 10 −10 metres or so in radius-a very surprising finding, as it implied a strong central charge less than 1/3000th of the diameter of the atom. įrom purely energetic considerations of how far particles of known speed would be able to penetrate toward a central charge of 100 e, Rutherford was able to calculate that the radius of his gold central charge would need to be less (how much less could not be told) than 3.4 × 10 −14 metres. In this May 1911 paper, Rutherford only committed himself to a small central region of very high positive or negative charge in the atom.įor concreteness, consider the passage of a high speed α particle through an atom having a positive central charge N e, and surrounded by a compensating charge of N electrons. In it, the atom is made up of a central charge (this is the modern atomic nucleus, though Rutherford did not use the term "nucleus" in his paper) surrounded by a cloud of (presumably) orbiting electrons. Rutherford presented his own physical model for subatomic structure, as an interpretation for the unexpected experimental results. Most of the beams went through the foil, but a few were deflected. If Thomson was correct, the beam would go straight through the gold foil. Rutherford designed an experiment to use the alpha particles emitted by a radioactive element as probes to the unseen world of atomic structure. 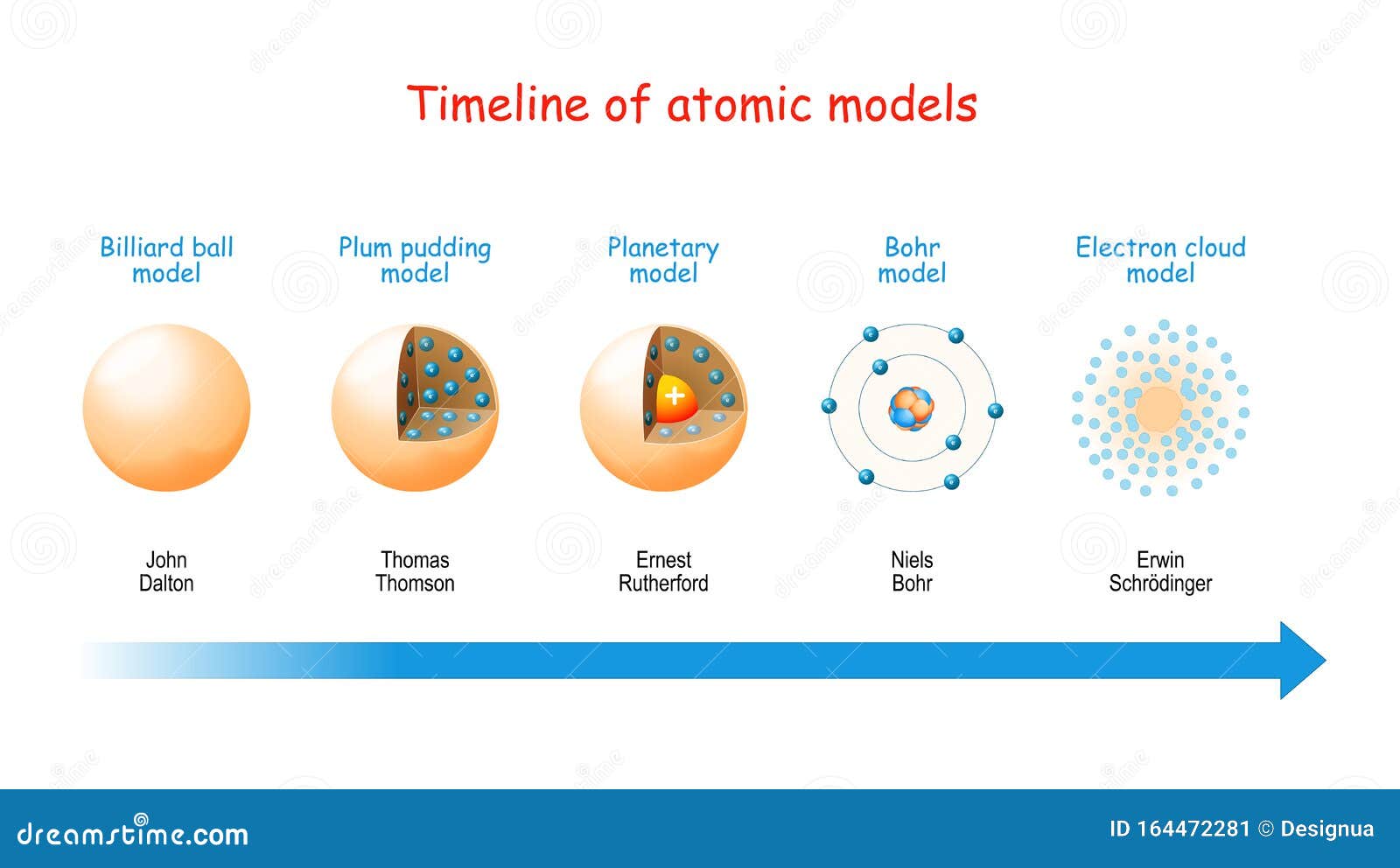
Rutherford overturned Thomson's model in 1911 with his well-known gold foil experiment in which he demonstrated that the atom has a tiny and heavy nucleus. The Rutherford model was subsequently superseded by the Bohr model.Įxperimental basis for the model Rutherford's new model for the atom, based on the experimental results, contained new features of a relatively high central charge concentrated into a very small volume in comparison to the rest of the atom and with this central volume containing most of the atom's mass this region would be known as the atomic nucleus. Thomson's plum pudding model of the atom was incorrect. Rutherford directed the Geiger–Marsden experiment in 1909, which suggested, upon Rutherford's 1911 analysis, that J. The Rutherford model was devised by Ernest Rutherford to describe an atom. 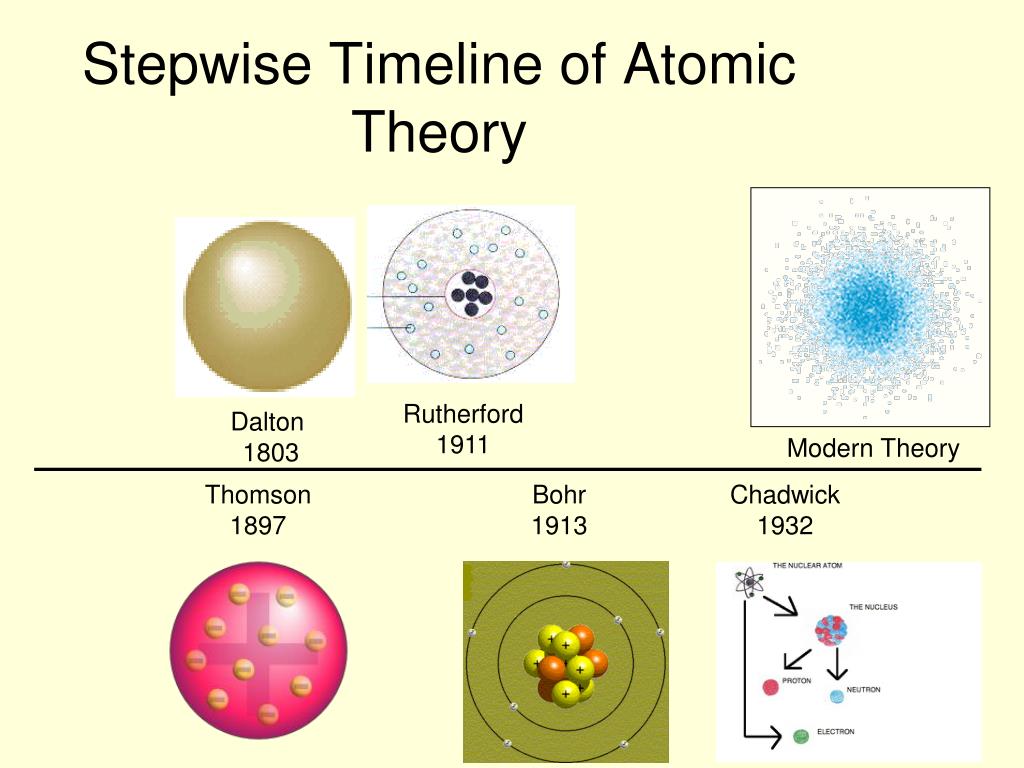
JSTOR ( June 2020) ( Learn how and when to remove this template message)īasic diagram of the atomic nuclear model: electrons in green and nucleus in red 3D animation of an atom incorporating the Rutherford model.Unsourced material may be challenged and removed. Please help improve this article by adding citations to reliable sources. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles.This article needs additional citations for verification.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
